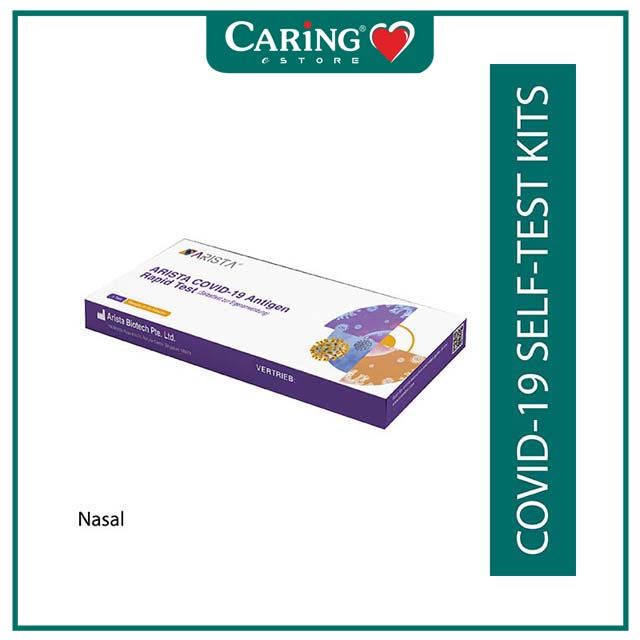
ARISTA COVID-19 ANTIGEN RAPID TEST 1S (RTK)
- Collect 1 point for every RM1 spent.
- Redeem RM1 with 150 points
- Birthday point EXTRA2x valid on whole birthday month.
GOLD18 POINTS
PLATINUM27 POINTS
-
FREE HOME DELIVERY
From RM150 (West Malaysia Only)
-
CLICK & PICK
Collect your parcel at nearest CARING PHARMACY branches
-
REWARD POINTS
With any purchase and redeem upon checkout
-
PAYMENT METHODS
Credit cards, e-wallets and more
The ARISTA™ COVID-19 Antigen Rapid Test is a lateral flow immunoassay intended for the qualitative in vitro detection of SARS-CoV-2 virus nucleocapsid (N)-protein antigen in humans. The test is intended as an aid in identifying COVID-19 infection and is available for professional use at point-of-care by healthcare providers or personnel trained in any laboratory and non-laboratory environment that meets the requirements, and for self-test use in accordance with local regulations.
Performance studies conducted on the ARISTA™ COVID-19 Antigen Rapid Test for professional use with nasopharyngeal specimens indicated a sensitivity of 99.4% and a specificity of 100% with overall agreement to PCR of 99.82%.
Performance studies conducted on the ARISTA™ COVID-19 Antigen Rapid Test for self-test use with anterior nasal specimens indicated a sensitivity of 95.8% and a specificity of 100% with overall agreement to PCR of 98.55%.
Performance studies conducted on the ARISTA™ COVID-19 Antigen Rapid Test for self-test use with deep throat sputum specimens indicated a sensitivity of 97.9% and a specificity of 100.00% with overall agreement to PCR of 99.26%.
Performance studies conducted on the ARISTA™ COVID-19 Antigen Rapid Test for self-test use with saliva specimens indicated a sensitivity of 95.8% and a specificity of 100.00% with overall agreement to PCR of 98.55%.
The ARISTA™ COVID-19 Antigen Rapid Test complies with European In-Vitro Diagnostic Devices Directive 98/79/EC, and was issued HSA Provisional Authorisation (MDPA2020-180) for supply in Singapore and export from Singapore.
Demo Video





![[HIJAB] NEUTROVIS 4PLY MASK HIJAB BLACK 50S](https://uat.caring2u.com/media/catalog/product/cache/e1a17c1de3163fefa238eb3bf35b1d95/1/0/10014064_EA_1.jpg)



![[KID] NEUTROVIS CH 4PLY MASK BLACK 50S](https://uat.caring2u.com/media/catalog/product/cache/e1a17c1de3163fefa238eb3bf35b1d95/1/0/10014065_EA_1.jpg)

The information below is required for social login
Sign In
Create New Account